Posts Tagged ‘contraception’
Live Session at APhA 2026
Lights, Camera, Access: Contraception Care at the Pharmacy Recent policy changes have expanded pharmacists’ ability to provide contraception care, including prescribing hormonal, nonhormonal, and emergency contraceptives. We will discuss the…
Read MoreIs it Safe for Birth Control Users to Go Years Without a Period?
Is it Safe for Birth Control Users to Go Years Without a Period? Conversations about contraceptive options have grown more frequent—and often more polarized—on social media platforms. Contraception is commonly used…
Read MoreComplex Contraception Cases – Live Webinar
Join us on Tuesday, November 18th at 5-6 pm PT / 8-9 pm ET In this live webinar we will evaluate complex contraception patient cases, identifying key factors that influence…
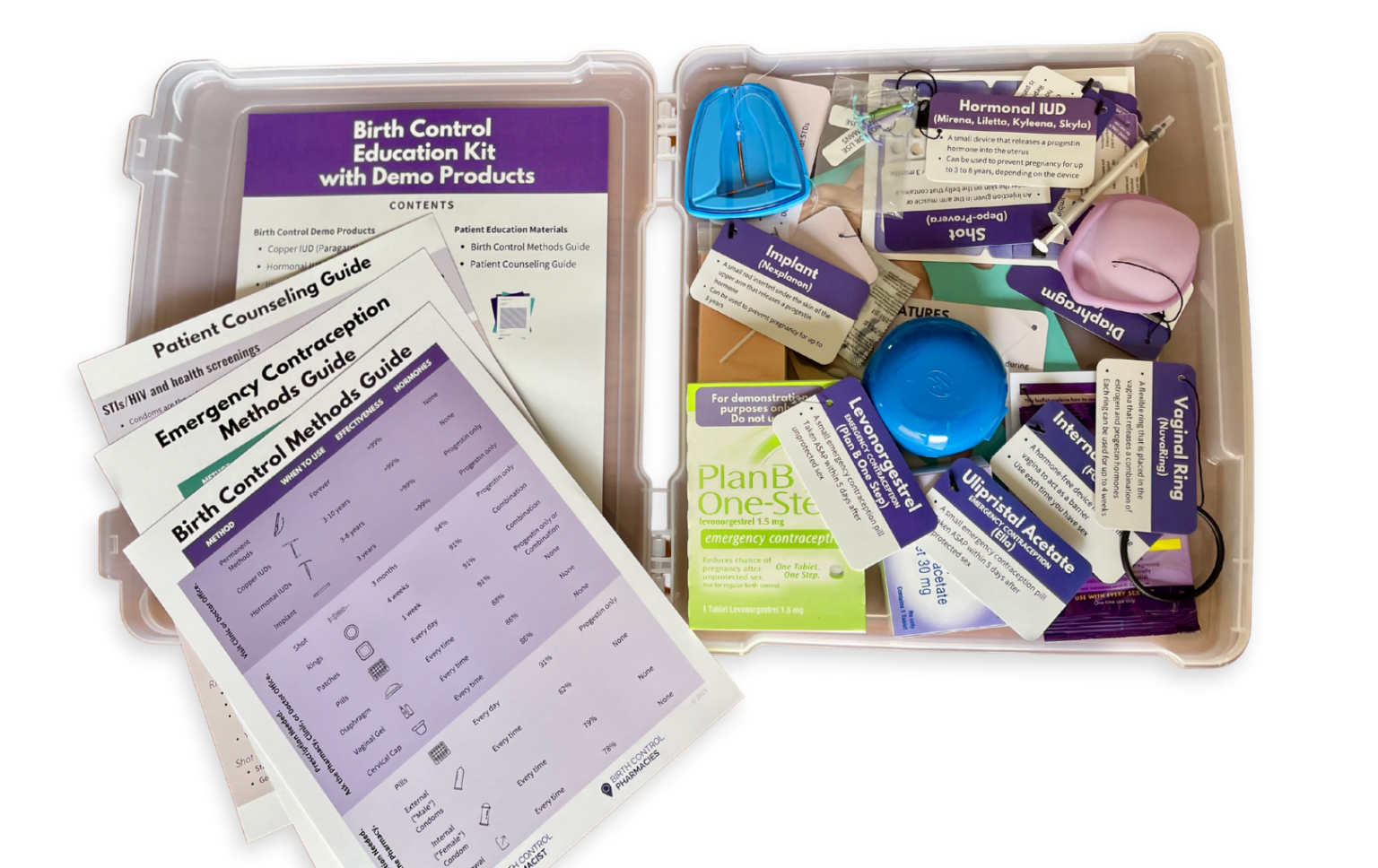
Read MoreEnhancing Patient Understanding With Contraceptive Demonstration Kits
Dr. Ashley Meredith discusses how she uses the Birth Control Education Kit as a useful resource for patient counseling and engaging classroom instruction.
Read MoreContinuing Contraception Care After Trump’s Orders
In light of recent actions over the last two weeks with our new administration, I wanted to provide updates and guidance to my pharmacist peers regarding reproductive health care. On…
Read MoreFounder Reflections on 2023
As I reflect on this past year, 2023 finally brought us some wins in reproductive health. And I personally had the opportunity to contribute to landmark steps forward. ⭐️ The…
Read MoreUpdated Report on State Policy Efforts to Expand Access to Contraception in Pharmacies
Download free 22-page report describing the current landscape of direct access to contraception in pharmacies, state policy approaches and experiences, as well as implementation.
Read MoreOutreach Initiative to Expand Access to Depot Medroxyprogesterone Acetate (DMPA)
New training program by Birth Control Pharmacist and Provide includes pharmacy best practices to combat stigma and communication guides and online resources to help connect patients with local resources.
Read MoreWebinar Introduces Pharmacists to New Hormonal Contraceptives
New training program by Birth Control Pharmacist and Provide includes pharmacy best practices to combat stigma and communication guides and online resources to help connect patients with local resources.
Read MoreWebinar Equips Pharmacists to Provide Contraception Care During COVID-19
New training program by Birth Control Pharmacist and Provide includes pharmacy best practices to combat stigma and communication guides and online resources to help connect patients with local resources.
Read More